The Carbon Cycle
Learning Objectives
By the end of this section, you will be able to do the following:
- Discuss the biological carbon cycle and why carbon is essential to all living things
Energy flows directionally through ecosystems, entering as sunlight (or inorganic molecules for chemoautotrophs) and leaving as heat during the many transfers between trophic levels. However, the matter that makes up living organisms is conserved and recycled. The six most common elements associated with organic molecules—carbon, nitrogen, hydrogen, oxygen, phosphorus, and sulfur—take a variety of chemical forms and may exist for long periods in the atmosphere, on land, in water, or beneath the Earth’s surface.
Carbon is one of the basic building blocks of life and the most abundant element in organisms, accounting for about half of typical dry biomass. Carbon is present in all organic molecules, and its role in the structure of macromolecules is of primary importance to living organisms. Carbon compounds contain especially high energy, particularly those derived from fossilized organisms, mainly plants, which humans use as fuel. Since the 1800s, the number of countries using massive amounts of fossil fuels has increased. Since the beginning of the Industrial Revolution, global demand for the Earth’s limited fossil fuel supplies has risen; therefore, the amount of carbon dioxide in our atmosphere has increased. This increase in carbon dioxide has been associated with climate change and other disturbances of the Earth’s ecosystems and is a major environmental concern worldwide. Thus, the “carbon footprint” is based on how much carbon dioxide is produced and how much fossil fuel countries consume.
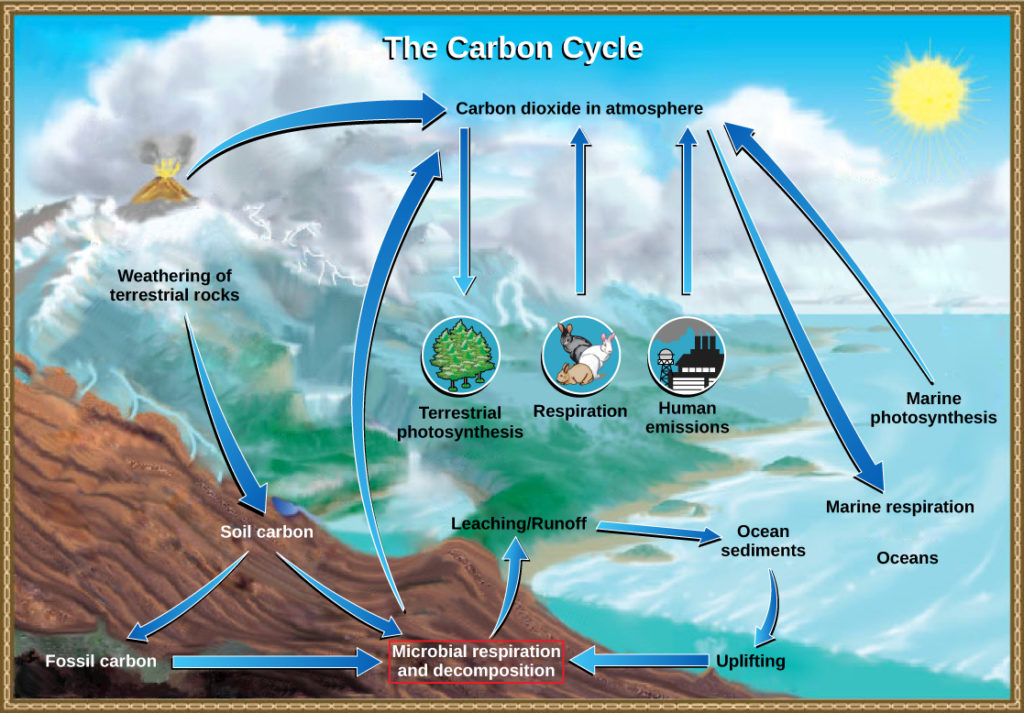
The carbon cycle is most easily studied as two interconnected sub-cycles: one dealing with rapid carbon exchange among living organisms and the other dealing with the long-term cycling of carbon through geologic processes. The entire carbon cycle is shown in Figure 1.
Click this link to read information about the United States Carbon Cycle Science Program.
The Biological Carbon Cycle
Living organisms are connected in many ways, even between ecosystems. A good example of this connection is the exchange of carbon between autotrophs and heterotrophs within and between ecosystems by way of atmospheric carbon dioxide.
Carbon dioxide is the basic building block that most autotrophs use to build multi-carbon, high-energy compounds, such as glucose. The energy harnessed from the sun is used by these organisms to form the covalent bonds that link carbon atoms together. These chemical bonds thereby store this energy for later use in the process of respiration. Most terrestrial autotrophs obtain their carbon dioxide directly from the atmosphere, while marine autotrophs may acquire it in the dissolved form (bicarbonate, HCO3⁻). However carbon dioxide is acquired, a by-product of the process is oxygen. Photosynthetic organisms are responsible for bringing the Earth’s atmosphere to the approximately 21 percent oxygen content that we observe today.
Heterotrophs and autotrophs are partners in biological carbon exchange (especially the primary consumers, largely herbivores). Heterotrophs acquire the high-energy carbon compounds from the autotrophs by consuming them, and breaking them down by respiration to obtain cellular energy, such as ATP. The most efficient type of respiration, aerobic respiration, requires oxygen obtained from the atmosphere or dissolved in water. Thus, there is a constant exchange of oxygen and carbon dioxide between the autotrophs (which need the carbon) and the heterotrophs (which need the oxygen). Gas exchange through the atmosphere and water is one way that the carbon cycle connects all living organisms on Earth.
The Biogeochemical Carbon Cycle
The movement of carbon through the land, water, and air is complex, and in many cases, it occurs much more slowly geologically than as seen between living organisms. Carbon is stored for long periods in what are known as carbon reservoirs, which include the atmosphere, bodies of liquid water (mostly oceans), ocean sediment, soil, land sediments (including fossil fuels), and the Earth’s interior.
As stated, the atmosphere is a major reservoir of carbon in the form of carbon dioxide and is essential to the process of photosynthesis. The level of carbon dioxide in the atmosphere is greatly influenced by the reservoir of carbon in the oceans. The exchange of carbon between the atmosphere and ocean influences how much carbon is found in each location, and each one affects the other reciprocally. Carbon dioxide (CO2) from the atmosphere dissolves in seawater, forming bicarbonate ions (HCO3⁻) which can be taken up and fixed by photosynthetic algae and bacteria, which are the base of the marine food web. More than 90 percent of the carbon in the ocean is found as bicarbonate ions. Some of these bicarbonate ions combine with seawater calcium to form calcium carbonate (CaCO3), the main mineral in marine organisms’ shells, and the primary component of coral reefs (the corals use calcium carbonate to build their stony skeletons). These organisms eventually form sediments on the ocean floor. Over geologic time, the calcium carbonate forms limestone, which comprises the largest carbon reservoir on Earth.
On land, carbon is stored in soil as a result of the decomposition of living organisms (by decomposers) or from weathering of terrestrial rock and minerals. This carbon can be leached into the water reservoirs by surface runoff. Deeper underground, on land and at sea, are fossil fuels: the anaerobically decomposed remains of plants that take millions of years to form. Fossil fuels are considered a nonrenewable resource because their use far exceeds their rate of formation. A non-renewable resource, such as fossil fuel, is either regenerated very slowly or not at all. Another way for carbon to enter the atmosphere is from land (including land beneath the surface of the ocean) by the eruption of volcanoes and other geothermal systems. Carbon sediments from the ocean floor are taken deep within the Earth by the process of subduction: the movement of one tectonic plate beneath another. Carbon is released as carbon dioxide when a volcano erupts or from volcanic hydrothermal vents.
Over almost all of geological time, the amount of CO2 absorbed by the global biota from the atmosphere was similar to that released through respiration and decomposition. Consequently, the cycling of this nutrient can be viewed as a steady-state system. In modern times, however, anthropogenic emissions (those caused by humans) have changed the atmospheric carbon balance. Global emissions of CO2 and CH4 are now larger than the uptake of these gasses, an imbalance that has resulted in increasing concentrations in the atmosphere. This phenomenon intensifies the greenhouse effect of Earth and results in a warming climate.
Animal husbandry by humans also increases atmospheric carbon. The large numbers of land animals raised to feed the Earth’s growing population results in increased carbon dioxide levels in the atmosphere due to farming practices and respiration and methane production. This is another example of how human activity indirectly affects biogeochemical cycles in a significant way. Although much of the debate about the future effects of increasing atmospheric carbon on climate change focuses on fossil fuels, scientists take natural processes, such as volcanoes and respiration, into account as they model and predict the future impact of this increase.
For an explanation of the cyclical relationship of carbon, humans and the environment, check out this animated lesson from TED-Ed:
Did I Get It?
CC Licensed Content, Shared Previously, Included in Ecology
- Biology 2e. Authors: Mary Ann Clark, Matthew Douglas and Jung Choi. Provided by: OpenStax CNX. Located at: Biology 2e. License: CC BY: Attribution 4.0.
- Biology for Majors II. Authors: Shelly Carter and Monisha Scott. Provided by: Lumen Learning. Located at: Biology for Majors II | Simple Book Production. License: CC BY: Attribution 4.0.
- Environmental Science: a Canadian perspective. Author: Bill Freedman. Provided by: Pressbooks. Located at: Environmental Science – Simple Book Publishing. License: CC BY-NC 4.0.

