Pleiotropy
Some alleles contribute to multiple phenotypes that may appear to be unrelated.
An example of this are mutations in the gene ATM. In humans and other animals, mutations in ATM cause ataxia (irregular movement), telangiectasias (blood vessel abnormalities visible on the surface of the eye), immune dysfunction, and a predisposition to certain kinds of cancer[1]. These phenotypes all seem pretty different, but they’re caused by mutations in the same gene.
Another example: in humans, mutations in the gene LMX1B cause Nail-Patella Syndrome. Individuals with Nail-Patella syndrome have abnormally shaped, underdeveloped, or absent fingernails or toenails. They also have skeletal abnormalities, including misshapen or missing patellae (kneecaps), and some people with Nail-Patella Syndrome develop kidney disease. The ATM and LMX1B disease-associated alleles are said to be pleiotropic, since the one allele causes many seemingly unrelated phenotypes.
A third example of pleiotropy is an allele of the gene that encodes the beta subunit of the protein hemoglobin. The HbS allele of the gene is linked with sickle cell disease (SCD). The healthy allele is called HbA. The HbS allele is most common among people with ancestors from sub-Saharan Africa and certain Mediterranean, Middle-Eastern, and Indian regions. In the United States, about 100,000 people are affected by sickle cell disease (about 3 in every 10,000)[2]. It affects 1 in every 365 Black or African American people.
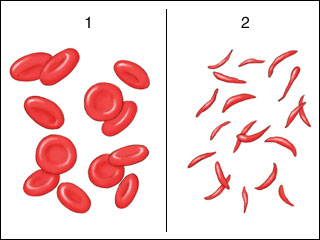
Individuals who are homozygous for the HbS allele have sickle cell disease, which gets its name from the abnormal shape of patients’ red blood cells, as seen under the microscope. Although healthy red blood cells are a flat disk shape with a depression in the center, under some conditions the red blood cells of patients with sickle cell disease form a crescent or sickle shape (Figure 9). The sickled red blood cells get stuck in capillaries, causing painful symptoms. Red blood cells of SCD patients are also fragile and short-lived compared to healthy cells, making patients anemic. SCD is also called sickle cell anemia.
The sickle shape is caused by a change in a single amino acid of the hemoglobin beta polypeptide: a single base difference in the DNA changes a glutamic acid in the healthy hemoglobin to a valine in the sickle cell hemoglobin. The hydrophobic valine makes the mutant hemoglobin somewhat insoluble in the aqueous environment of the cell, resulting in the clumping together of individual molecules of the mutant hemoglobin. This causes the cells to deform and sickle.
But the HbS allele also confers resistance to the disease malaria: individuals who are heterozygous for the HbS allele are far less likely to develop malaria if they are exposed to the infectious parasite that causes the disease. In this case, the two phenotypes of sickle cell disease and resistance to malaria make the allele pleiotropic.
As with penetrance and expressivity, the underlying mechanism for pleiotropy may not be understood. However, in many cases it is because one single gene has many downstream targets. In the example of ATM, the encoded protein is a kinase that adds a phosphate group to many target proteins. The phosphate group changes the activity of the target proteins, each of which is involved in a different cellular process. The altered activity of the downstream target proteins leads to the different mutant phenotypes.
For the HbS allele: resistance to malaria is likely a side effect of the sickling, since the malarial parasite P. falciparum infects red blood cells. Although individuals heterozygous for the HbS allele do not have sickle cell disease, their hemoglobin can clump and their cells can sickle under conditions of low oxygenation. One hypothesis is that the sickling that occurs in low-oxygen conditions interferes with the parasite’s life cycle, preventing symptoms of malaria from occurring[3].
Test Your Understanding
Media Attributions
- Sickle cell anemia © Wikipedia is licensed under a CC0 (Creative Commons Zero) license
- Online Mendelian Inheritance in Man, OMIM®. Johns Hopkins University, Baltimore, MD. MIM Number: #208900: last edit 03/22/2022. World Wide Web URL: https://omim.org/. ↵
- Data & Statistics on Sickle Cell Disease | CDC. Centers for Disease Control and Prevention https://www.cdc.gov/ncbddd/sicklecell/data.html (2022). ↵
- Archer, N. M. et al. Resistance to Plasmodium falciparum in sickle cell trait erythrocytes is driven by oxygen-dependent growth inhibition. Proceedings of the National Academy of Sciences 115, 7350–7355 (2018). ↵

